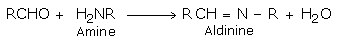Nucleophilic Addition Reactions
Nucleophilic Addition Reactions
Being unsaturated, aldehydes and ketones undergo addition reactions. They have a polar carbonyl group. The carbon of the carbonyl group being electrophilic, is readily attacked by the nucleophiles. Hence the most typical reactions of aldehydes and ketones are nucleophilic addition reactions to carbon-oxygen double bond.
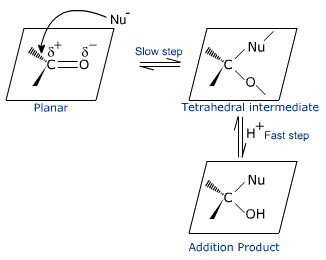
Mechanism of nucleophilic addition
The nucleophile (Nu-) attacks the carbonyl carbon from above or below the plane of the carbonyl group leading to a C-Nu bond formation.
This is accompanied by heterophilic cleavage of the weaker carbon-oxygen pi-bond. The electron pair of the pi-bond is completely transferred to the oxygen atom. The electronegative oxygen atom thus gets a negative charge. During this process, the hybridization of the carbonyl carbon changes from trigonal to tetrahedral and the oxygen atom is pushed out of the plane of the carbonyl group.
The negatively charged tetrahedral intermediate is basic and captures, a proton from the medium to give the electrically neutral product. The net result is addition of Nu- and H+ across the carbon-oxygen double bond.
Relative Reactivity of Aldehydes and Ketones
In general ketones are less reactive than aldehydes on account of the following facts.
(i) Electron releasing effect
In the mechanism of nucleophilic addition, the first step involving the formation of the tetrahedral intermediate, is accelerated by electron withdrawing groups and retarded by electron donating groups.
In ketones, the carbonyl carbon is attached to two alkyl groups, which are electron releasing in nature. These alkyl groups push electrons towards carbonyl carbon and thus decrease the magnitude of positive charge on it and make it less susceptible to nucleophilic attack. In an aldehyde there is only one electron donating group as against two in ketones.
(ii) Steric effect
The tetrahedral intermediate is more crowded when bulkier groups are attached to carbonyl carbon. In ketones the presence of two bulky alkyl groups hinders the approach of the nucleophile to the carbonyl carbon. This factor is called the steric factor.
Some important examples of nucleophilic addition reactions.
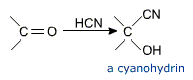
a) Addition of hydrogen cyanide (HCN). HCN adds to aldehydes and ketones to form cyanohydrins.
Cyanohydrins are useful synthetic intermediates.
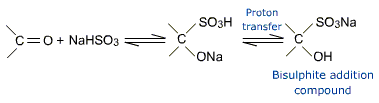
b) Addition of sodium bisulphite (NaHSO3)
NaHSO3 adds to aldehydes and ketones to form crystalline addition products.
The bisulphite addition compound can be converted back to the original carbonyl compound by treating it with dilute mineral and or alkali. Therefore the bisulphite addition compounds are useful for separation and purification of aldehydes.
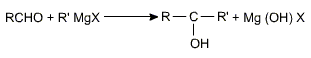
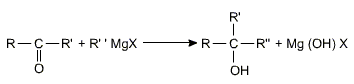
c) Addition of Grignard reagents
Aldehydes and ketones add on to Grignard's reagents to form alcohols on hydrolysis.
Grignard's reagents gives a primary alcohol with HCHO, a secondary alcohol is produced with any other aldehyde and a tertiary alcohol with a ketone.
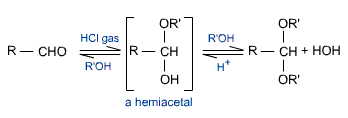
d) Addition of alcohols
Aldehydes react with alcohols in the presence of dry HCl gas to form acetals a gem-dialkoxy compound. In this reaction, the addition of one molecule of alcohol to one molecule of aldehyde results in the formation of an alkoxy alcohol intermediate known as a hemiacetal. A hemiacetal contains both an ether as well as alcohol functional groups. It is an unstable compound and cannot be isolated. It further reacts with alcohol to form stable acetal.
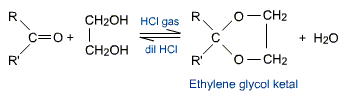
Ketones react with ethylene glycol under similar conditions to form cyclic products known as ethylene glycol ketals.
Dry hydrogen chloride absorbs the water produced in these reactions and drives the equilibrium in the forward direction. Acetals and Ketals are hydrolyzed with aqueous mineral acids.
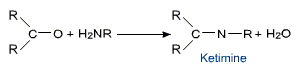
e) Addition of ammonia and its derivatives
Nitrogen nucleophiles such as ammonia and its derivatives H2N - Z add to carbonyl group of aldehydes and ketones. The reaction is reversible and catalyszed by acid. The equilibrium favors the product formation due to rapid dehydration of the tetrahedral addition product.
The net result is replacement of the >C = O group with >C = N - Z group.
Aldehydes and ketones react with primary amines to form Schiff's base. These compounds are also called imines.
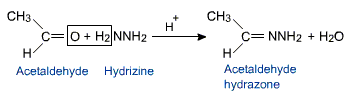
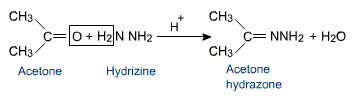
Aldehydes and ketones react with hydrazine to form hydrazones.
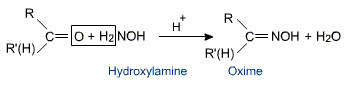
Aldehydes and ketones react with hydroxylamine to form oximes.
The oximes can be hydrolyzed back to aldehydes and ketones by reaction with acids.
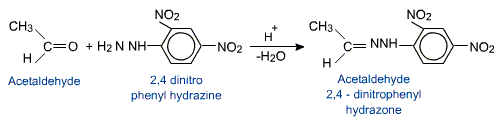
Aldehydes and ketones react with 2,4-dinitrophenyl hydrazine to form
2,4-dinitrophenylhydrazones commonly known as DNP or Brady's reagent.
2,4-DNP derivatives are yellow, orange or red solids useful for characterization of aldehydes and ketones.
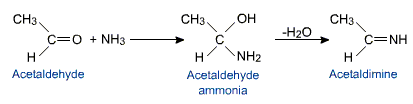
Like ammonia derivatives ammonia also reacts with aldehydes (except formaldehyde) and ketones to form the products called imines.
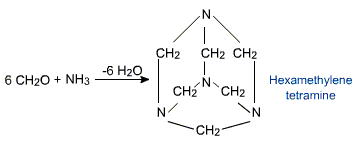
However formaldehyde reacts with ammonia to form hexamethylene tetramine (CH2)6N4 also known asurotropine.
Urotropine is used as a medicine to treat urinary infections. Nitration of urotropine under controlled condition gives an explosive RDX (Research and development explosive).
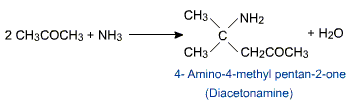
Acetone reacts with ammonia to form diacetonamine.
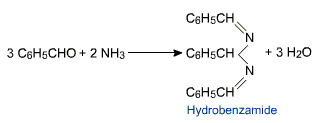
Unlike aliphatic aldehydes and ketones benzaldehyde on reaction with ammonia produces a complex product hydrobenzamide.
Formalin and its uses
Methanol is extremely soluble in water and its 40% aqueous solution is called formalin. Formalin is a disinfectant and a preservative for biological samples. Methanol is used in the manufacture of bakelite, resins and other polymers.
 SureDen
SureDen