Kossel Lewis Approach
Chemical Bond is a force that acts between 2 or more atoms to hold them together as a stable molecule.
Cause of chemical combination=>Lewis- approach=>Kossel Lewis octet rule=>It stats that the atoms of different elements combine with each other in order to complete their respective octets (i:e & Ä“ in outermost shell) or duplet (i.e. outermost shell having 2 Ä“) in case of H, Li & Be, to attain nearest noble gas configuration .
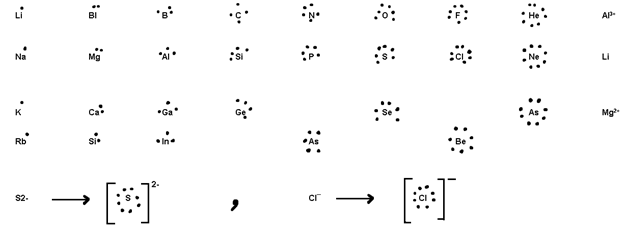
Lewis dot symbols =>The symbol of atom is surrounded by dots equal to the no. of valence electrons.eg
Lewis dot symbols of =>
Models of Chemical Combination:=>
- Electrovalent or Ionic bond =>It’s formed by complete transference of one or more electrons from one atom to another.
- By sharing of electrons :-
- Covalent bond:-It is formed by mutual sharing of electrons.
- Coordinate or dative bond:-It’s formed by one sided sharing of electrons.
Related Keywords
 SureDen
SureDen